Triruthenium dodecacarbonyl
| Triruthenium dodecacarbonyl | |
|---|---|
|
cyclo-tris(tetracarbonylruthenium) |
|
|
Other names
Ruthenium carbonyl |
|
| Identifiers | |
| CAS number | 15243-33-1 |
| Properties | |
| Molecular formula | C12O12Ru3 |
| Molar mass | 639.33 g/mol |
| Appearance | orange solid |
| Density | 2.48 g/cm3 |
| Melting point |
224 °C |
| Boiling point |
sublimes in vacuum |
| Solubility in water | insoluble |
| Solubility in organic solvents | soluble |
| Structure | |
| Molecular shape | D3h cluster |
| Dipole moment | 0 D |
| Hazards | |
| Main hazards | Toxic |
| Related compounds | |
| Related compounds | Triiron dodecacarbonyl Triosmium dodecacarbonyl |
| (verify) (what is: /?) Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) |
|
| Infobox references | |
Triruthenium dodecacarbonyl is the chemical compound with the formula Ru3(CO)12. This orange-colored metal carbonyl cluster is a precursor to other organoruthenium compounds.
Contents |
Structure and synthesis
The cluster has D3h symmetry, consisting of an equilateral triangle of Ru atoms, each of which bears two axial and two equatorial CO ligands.[1] Os3(CO)12 has the same structure, whereas Fe3(CO)12 is different, with two bridging CO ligands, resulting in C2v symmetry.
Ru3(CO)12 is prepared by treating solutions of ruthenium trichloride with carbon monoxide, usually under high pressure.[2][3] The stoichiometry of the reaction is uncertain, one possibility being the following:
- 6 RuCl3 + 33 CO + 18 CH3OH → 2 Ru3(CO)12 + 9 CO(OCH3)2 + 18 HCl
Reactions
The chemical properties of Ru3(CO)12 have been widely studied, and the cluster has been converted to hundreds of derivatives. High pressures of CO convert the cluster to the monomeric pentacarbonyl, which reverts back to the parent cluster upon standing.
- Ru3(CO)12 + 3 CO
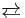 3 Ru(CO)5 Keq = 3.3 x 10-7 mol dm–3 at room temperature
3 Ru(CO)5 Keq = 3.3 x 10-7 mol dm–3 at room temperature
The instability of Ru(CO)5 contrasts with the robustness of the corresponding Fe(CO)5. The condensation of Ru(CO)5 into Ru3(CO)12 proceeds via initial, rate-limiting loss of CO to give the unstable, coordinatively unsaturated species Ru(CO)4. This tetracarbonyl binds Ru(CO)5, initiating the condensation.[4]
Upon warming under a pressure of hydrogen, Ru3(CO)12 converts to the tetrahedral cluster H4Ru4(CO)12.[5] Ru3(CO)12 undergoes substitution reactions with Lewis bases:
- Ru3(CO)12 + n L → Ru3(CO)12-nLn + n CO (n = 1, 2, or 3)
where L is a tertiary phosphine or an isocyanide.
Ru-carbido clusters
At high temperatures, Ru3(CO)12 converts to a series of clusters that contain interstitial carbido ligands. These include Ru6C(CO)17 and Ru5C(CO)15. Anionic carbido clusters are also known, including [Ru5C(CO)14]2- and the bioctahedral cluster [Ru10C2(CO)24]2-.[6]
Ru3(CO)12 -derived carbido compounds have been used to synthesize nanoparticles for catalysis. These particles consist of 6-7 atoms and thus are all surface, resulting in extraordinary activity.
References
- ^ Slebodnick, C.; Zhao, J.; Angel, R.; Hanson, B. E.; Song, Y.; Liu, Z.; Hemley, R. J., "High Pressure Study of Ru3(CO)12 by X-ray Diffraction, Raman, and Infrared Spectroscopy", Inorg. Chem., 2004, 43, 5245-52. doi:10.1021/ic049617y
- ^ Bruce, M. I.; Jensen, C. M.; Jones, N. L. “Dodecacarbonyltriruthenium, Ru3(CO)12” Inorganic Syntheses, 1989, volume 26, pages 259-61. ISBN 0-471-50485-8.
- ^ M. Faure, C. Saccavini, G. Lavigne “Dodecacarbonyltriruthenium, Ru3(CO)12” Inorganic Syntheses, 2004 Vol 34, p. 110. ISBN 0-471-64750-0.
- ^ Hastings, W. R.; Roussel, M. R.; Baird, M. C. “Mechanism of the conversion of [Ru(CO)5] into [Ru3(CO)12]” Journal of the Chemical Society, Dalton Transactions, 1990, pages 203-205. DOI: 10.1039/DT9900000203
- ^ Bruce, M. I.; Williams, M. L. “Dodecacarbonyl(tetrahydrido)tetraruthenium, Ru4(μ-H)4(CO)12” Inorganic Syntheses, 1989, volume 26, pages 262-63. ISBN 0-471-50485-8.
- ^ Nicholls, J. N.; Vargas, M. D. “Carbido-Carbonyl Ruthenium Cluster Complexes” Inorganic Syntheses, 1989, volume 26, pages 280-85. ISBN 0-471-50485-8ISBN.
|
|||||